Immunotherapy has the potential to treat a wide variety of conditions, from autoimmune disease to cancer. However, the traditional immunization approach to therapeutic antibody discovery is severely limited in scale and throughput, making the development of antibodies targeting key receptor proteins both time-consuming and resource-intensive.
High-quality synthetic antibody libraries can help overcome these hurdles by providing researchers with access to millions of antibodies that display a natural spectrum of variation, enabling rapid screening and optimization.
Twist Biopharma, a division of Twist Bioscience, was built to do exactly that.
By leveraging Twist Bioscience’s world-leading oligonucleotide synthesis platform to build high-quality antibody discovery libraries, Twist Biopharma is accelerating therapeutic discovery and development in many areas, including hard-to-drug targets.
Emphasizing this point, Twist Biopharma recently announced the discovery and preclinical validation of optimized, fully-human, highly-potent antibody leads targeting two hard-to-drug receptors, ADORA2A and GLP1R.
Therapeutic antibodies and hard-to-drug targets
With Twist Bioscience’s ability to generate millions of precisely written oligonucleotides, Twist Biopharma is able to build out antibody libraries that focus on specific target types, such as receptor proteins.
Ready access to these libraries not only accelerates antibody discovery, it also makes the discovery process much easier.
Take ADORA2A as an example. This protein operates as an adenosine receptor and immune checkpoint protein that prevents the inappropriate activation of T-cells and is an interesting immuno-oncology target.
Unfortunately, in the context of cancer, the normal, immunosuppressive role of a checkpoint protein is counterproductive, as it can prevent a T-cell from destroying a tumor cell. If there were a checkpoint inhibitor that blocked ADORA2A activity, it would allow cytotoxic—effectively allowing the patient’s immune system to fight back and destroy cancer.
Immune checkpoint blockades are one of the newest pillars of cancer therapeutic development. Current examples in the literature for targeting ADORA2A involve small molecule antagonists (ref 1). Selective antibody binders of ADORA2A are therefore a largely untapped, exciting immunotherapeutic candidate.
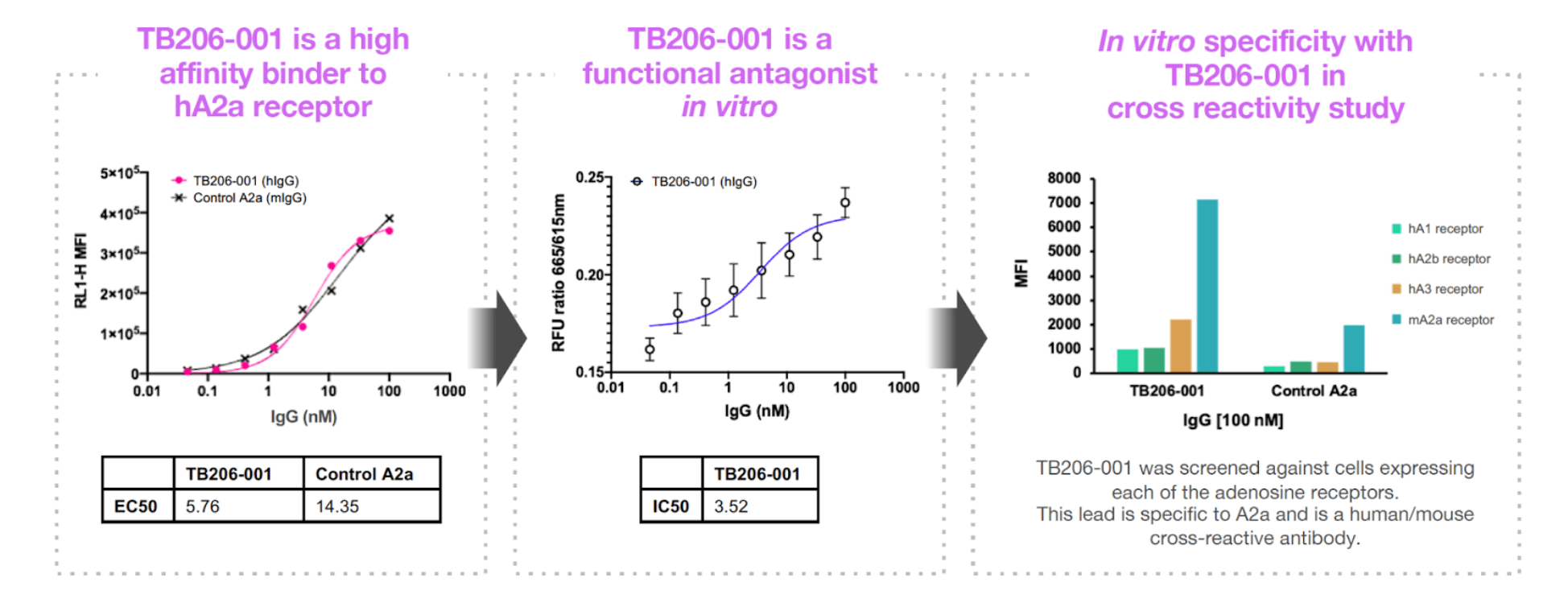
Using its proprietary biologics discovery and optimization platform, Twist Biopharma identified a potent high-affinity antibody, TB206-001, amongst other promising leads. Initial in vitro assays show that TB206-001 binds with high affinity to both human and mouse ADORA2A and has an IC50 that is 15-fold lower than a currently available, potent small-molecule inhibitor of ADORA2A, ZM-241,385 (ref 2). Subsequent in vivo testing also demonstrated this lead’s efficacy in animal models of cancer.
Twist believes TB206-001 is now the first high-affinity antagonistic anti-ADORA2A antibody to to demonstrate preclinical activity.
Twist Biopharma also recently produced therapeutic antibody candidates against GLP1R, a receptor involved in the regulation of insulin secretion (ref 3). Amongst its libraries, Twist Biopharma identified potent, high-affinity agonistic and antagonistic antibodies targeting GLP1R, both of which are now poised for further development as potential immunotherapies for obesity and hypoglycemia, respectively. (GLP1R is also a target for the treatment of diabetes and metabolic disorders.)
🔎 Looking for the data?
- Preclinical data on Twist Biopharma’s group of GLP1R antibodies was recently published in a peer-reviewed article in mAbs.
- Information and resources on these two antibody targets can be found on our clinical development and out-licensing page.
- You can also listen to Twist Bioscience’s Chief Scientific Officer, Dr. Aaron Sato, discuss next-generation synthetic antibody libraries, kinetic assays, and high throughput screening in this webinar. Click here to watch.
Oligonucleotide synthesis accelerates therapeutic antibody discovery
Our high throughput oligonucleotide synthesis technology allows Twist Biopharma to rapidly build antibody discovery libraries from large volumes of oligonucleotides. By synthesizing oligonucleotides with base-by-base precision, we can build antibody libraries to include only the antibody sequences naturally occurring in the human body. Doing so minimizes toxicity, improves pharmacokinetic properties, and saves development time.
What makes our oligonucleotide synthesis platform—and thus Twist Biopharma—so powerful is our explicit capability to avoid randomization (e.g. NNK bases or error-prone PCR) in the library construction process. Instead, we precisely code variation into oligonucleotide sequences using our silicon-based synthesis process. Because of this, Twist Biopharma’s libraries exhibit tight uniformity, minimal bias, and even variant representation—properties that have allowed for miniaturization and automation of the Twist Biopharma phage display-driven discovery pipeline.
These antibody libraries are also flexible. They can be built into various antibody scaffolds, such as IgG, scFv, Fab, and VHH, making Twist Biopharma uniquely situated to discover antibody candidates against hard-to-drug targets.
Working with Twist Biopharma on antibody engineering, discovery, and development
Twist Biopharma’s anti-ADORA2A and GLP1R antibody candidates are available for licensing and further development. In addition, Twist Biopharma offers a suite of antibody discovery and optimization capabilities for partners looking to identify and optimize biologics of all modalities against specific disease targets.
Over the course of the coming year, Twist Biopharma plans to generate new high-quality libraries, as well as develop new antibody therapies against hard-to-drug targets.
Learn more about Twist Biopharma here. If your company is interested in a partnership or licensing agreement, please contact Twist Biopharma at biopharma@twistbioscience.com.
References
- Adenosine 2A Receptor Blockade as an Immunotherapy for Treatment-Refractory Renal Cell Cancer. Fong L, et al., 2020. Cancer Discov. January 1 2020, 10:1, 40-53, DOI: 10.1158/2159-8290.CD-19-0980
- Tumor Immunotherapy Using A2A Adenosine Receptor Antagonists. Zhang J, et al., September 8 2020. Pharmaceuticals. 13:237, DOI: 10.3390/ph13090237
- Functional GLP-1R antibodies identified from a synthetic GPCR-focused library demonstrate potent blood glucose control. Liu Q, et al., March 12 2021. mAbs. 13:1, DOI: 10.1080/19420862.2021.1893425
Recommended Resources
- Terms & Conditions
- Policy on Unsolicited Submissions of Information
- Privacy Policy
- Regulatory and Quality Information
- Security
- Payment Information
-
© 2025 Twist Bioscience. All rights reserved.